Does Atogepant Offer a Safe and Efficacious Option for Episodic Migraine Prophylaxis? A Systematic Review and Meta-analysis
Article information
Abstract
Migraine, a chronic neurological disorder, imposes a significant burden on individuals and healthcare systems globally. This systematic review and meta-analysis evaluated the efficacy and safety of atogepant in preventing episodic migraine (EM) in adults. A systematic search was conducted in four major databases (PubMed, Scopus, Web of Science, and Cochrane CENTRAL) up to June 2024. The inclusion criteria targeted randomized controlled trials (RCTs) comparing atogepant to placebo or standard care in patients with EM. Statistical analyses were performed using Review Manager (RevMan) software. Four RCTs with 2,018 patients receiving atogepant and 761 patients receiving placebo or standard care were included. Atogepant significantly reduced monthly migraine days compared to placebo at 10 mg daily (mean difference [MD], –1.16 days; 95% confidence interval [95% CI], –1.60 to –0.73), 30 mg daily (MD, –1.15 days; 95% CI, –1.64 to –0.66), 60 mg daily (MD, –1.48 days; 95% CI: –2.36 to –0.61 days), 30 mg twice daily (MD, –1.30 days; 95% CI, –2.17 to –0.43), and 60 mg twice daily (MD, –1.20 days; 95% CI, –1.90 to –0.50). A ≥50% reduction in migraine days was frequently significantly achieved with atogepant across all dosages. Atogepant was generally well tolerated, though it was associated with higher incidence rates of constipation and nausea compared to placebo. Atogepant is an effective and well-tolerated option for preventing EM, offering patients a noninvasive oral alternative to injectable therapies. Further research is warranted to explore its long-term safety and efficacy in diverse patient populations and refine its role in this field.
INTRODUCTION
Migraine is a prevalent neurological disorder that significantly impacts quality of life. Episodic migraine (EM), characterized by headache attacks occurring fewer than 15 days per month, often imposes a substantial burden on patients, particularly when attacks are frequent and inadequately managed.1,2 Despite advances in acute treatments, many patients continue to experience recurrent migraines, highlighting the need for effective preventive options.3,4
Currently available preventive therapies, such as beta-blockers, anticonvulsants, and calcium channel blockers, often have limitations related to efficacy and tolerability.5,6 These challenges and the lack of migraine-specific mechanisms in older treatments underscore the necessity for targeted approaches.
Recent advancements in understanding migraine mechanisms have identified calcitonin gene-related peptide (CGRP) as a pivotal target in migraine pathogenesis. Elevated CGRP levels during migraine attacks contribute to vasodilation and neurogenic inflammation, processes central to migraine development.7,8 Consequently, the development of CGRP antagonists, including both injectable monoclonal antibodies (e.g., fremanezumab, eptinezumab, galcanezumab) and small-molecule CGRP receptor antagonists known as “gepants” (e.g., atogepant), has revolutionized preventive migraine treatment.9 Among these, atogepant, an orally active CGRP receptor antagonist, represents a novel approach to EM prevention, offering the advantages of targeting specific pathophysiological mechanisms and accommodating patient preferences for oral administration.10,11 Its oral formulation addresses a key patient preference for non-invasive treatment options, especially in comparison to injectables, which, despite their efficacy, may pose compliance challenges.11
Initial clinical studies have shown that atogepant significantly reduces the frequency of migraine attacks in patients with EM.12-15 However, the conclusions from individual trials are often limited by factors such as small sample sizes, differences in study designs, and variation in outcome measures. Therefore, a comprehensive meta-analysis is warranted to aggregate data across studies, offering a more precise evaluation of atogepant’s efficacy and safety profile. This meta-analysis addresses these gaps by systematically evaluating the efficacy and safety of atogepant, offering robust evidence to support its role in EM prevention.
By systematically assessing atogepant’s therapeutic potential, this review seeks to contribute to clinical decision-making and optimize the management of migraine, particularly for patients inadequately served by existing preventive treatments. Furthermore, it aims to highlight the limitations of previous analyses and clarify atogepants’ efficacy and safety across diverse patient populations, guiding their future role in personalized migraine care.
METHODS
This systematic review and meta-analysis followed rigorous methodology as outlined in the ‘Cochrane Handbook for Systematic Reviews of Interventions’16 and adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines17 to ensure transparency and reproducibility.
1. Search strategy and screening
A comprehensive search of four databases (PubMed, Scopus, Web of Science, and Cochrane CENTRAL) was performed up to June 5, 2024, using the search query: [(“atogepant” OR “calcitonin gene-related peptide antagonists” OR “CGRP antagonists”) AND (“migraine disorder” OR “chronic migraine” OR “episodic migraine” OR “headache disorders”)]. No filters were applied to ensure a broad capture of relevant studies. The search strategy was designed to identify randomized controlled trials (RCTs) comparing atogepant with placebo or standard care in patients diagnosed with EM. We aimed to include all RCTs, whether open-label or double-blinded, to provide a comprehensive evaluation of atogepant’s efficacy and safety profile in EM prevention. The inclusion of an open-label study was justified explicitly by its relevance to safety data and long-term outcomes, which complemented the controlled trial data and addressed existing gaps in the literature. This approach ensured a holistic and comprehensive review of all available evidence regarding atogepant.
Two independent reviewers (A.M. and M.E.M.) screened titles and abstracts using Rayyan software,18 with discrepancies resolved by consensus and arbitration from a third reviewer (A.M.A.). Studies that met the inclusion criteria progressed to full-text screening, and any conflicts were further discussed to reach a final decision.
2. Data extraction
Two reviewers used Microsoft Excel 2021 (Microsoft) to extract data independently, ensuring accuracy and completeness. Extracted data included:
• Study characteristics: study design, sample size, country, duration, inclusion criteria, and key findings.
• Patient characteristics: demographics such as age, sex, body mass index, and migraine duration.
• Risk of bias domains as outlined by the revised Cochrane risk-of-bias tool (RoB-2).
• Efficacy outcomes: changes in monthly migraine days, headache days, and acute medication use days, along with the proportion of patients achieving a ≥50% reduction in monthly migraine days.
• Safety outcomes: adverse events (AEs) such as upper respiratory tract infections (URTIs), nausea, constipation, nasopharyngitis, urinary tract infections (UTIs), and fatigue, as well as serious adverse events (SAEs), treatment-related AEs, and discontinuations due to AEs. Discrepancies in data extraction were resolved through discussion or consultation with a third reviewer.
3. Risk of bias assessment
Two authors independently assessed the risk of bias in the included studies using the RoB-2.19 This tool evaluates bias across five domains: randomization, deviations from intended interventions, missing outcome data, measurement of outcomes, and reporting bias. Each domain was rated as low risk, some concerns, or high risk. If any domain showed a high risk or multiple domains showed concerns, the study was considered at high risk of bias.
4. Statistical analysis
Data were analyzed using Review Manager (RevMan) software.20 Continuous outcomes (e.g., monthly migraine days, headache days, and acute medication use days) were summarized as mean differences (MD) with 95% confidence intervals (CIs). For dichotomous outcomes (e.g., ≥50% reduction in migraine days and AEs), risk ratios (RR) or risk differences (RD) were calculated with 95% CI.
Heterogeneity was assessed using the chi-square test, with the extent of heterogeneity measured using the I-squared (I2) statistic. A chi-square p-value less than 0.1 or an I2 greater than 50% indicated significant heterogeneity. In cases of significant heterogeneity, a random-effects model was used; otherwise, a fixed-effects model was applied.18
Subgroup analyses were conducted to handle significant heterogeneity and evaluate the efficacy of different atogepant dosage levels (10 mg once-daily [QD], 30 mg QD, 60 mg QD, 30 mg twice-daily [BID], and 60 mg BID) on primary outcomes. Additionally, an overall analysis combining all dosage groups was performed, following the Cochrane Handbook’s recommended formula.21 Given the limited number of included studies (fewer than 10), publication bias could not be formally assessed using funnel plots.22
RESULTS
1. Search and screening
The systematic search across four databases yielded 1,598 articles. After removing duplicates, 951 unique records were identified. Title and abstract screening narrowed these to 28 studies, and after full-text evaluation, four RCTs13-15,23 met the inclusion criteria for this meta-analysis (Figure 1).
2. Baseline characteristics
The included trials involved a total of 2,018 patients treated with atogepant and 761 patients in placebo or standard care groups. The mean age across the studies was 41.3 years, with 312 males among the participants. Three of the four trials were double-blinded and multicenter in design, except for Ashina et al.,14 which was not double-blinded and compared atogepant to standard care. The latter one was included in this systematic review, not the analysis to allow for consistent analysis of atogepant versus placebo. All studies followed the diagnostic criteria for EM as defined by the International Classification of Headache Disorders, 3rd edition.24 Study details are provided in Table 1.
3. Risk of bias assessment
The ROB-2 tool was used to assess the risk of bias, and all studies demonstrated a low risk. Each trial adequately implemented randomization procedures, and no significant deviations from the intended interventions were observed. Details of the risk-of-bias assessment are included in Supplementary Figure 1 (available online).
4. Mean difference in monthly migraine days
Three studies13,15,23 reported data on monthly migraine days across varying atogepant doses (10 mg QD, 30 mg QD, 60 mg QD, 30 mg BID, and 60 mg BID).
• At 10 mg QD and 30 mg QD dosages: In two studies, atogepant at 10 mg QD significantly reduced monthly migraine days compared to placebo (MD: –1.16 days, 95% CI: –1.60 to –0.73, p<0.00001), with no significant heterogeneity (p=0.83, I2=0%). Similarly, at 30 mg QD, there was a significant reduction in migraine days (MD: –1.15 days, 95% CI: –1.64 to –0.66, p<0.00001), with low heterogeneity (p=0.21, I2=36%) (Figure 2).
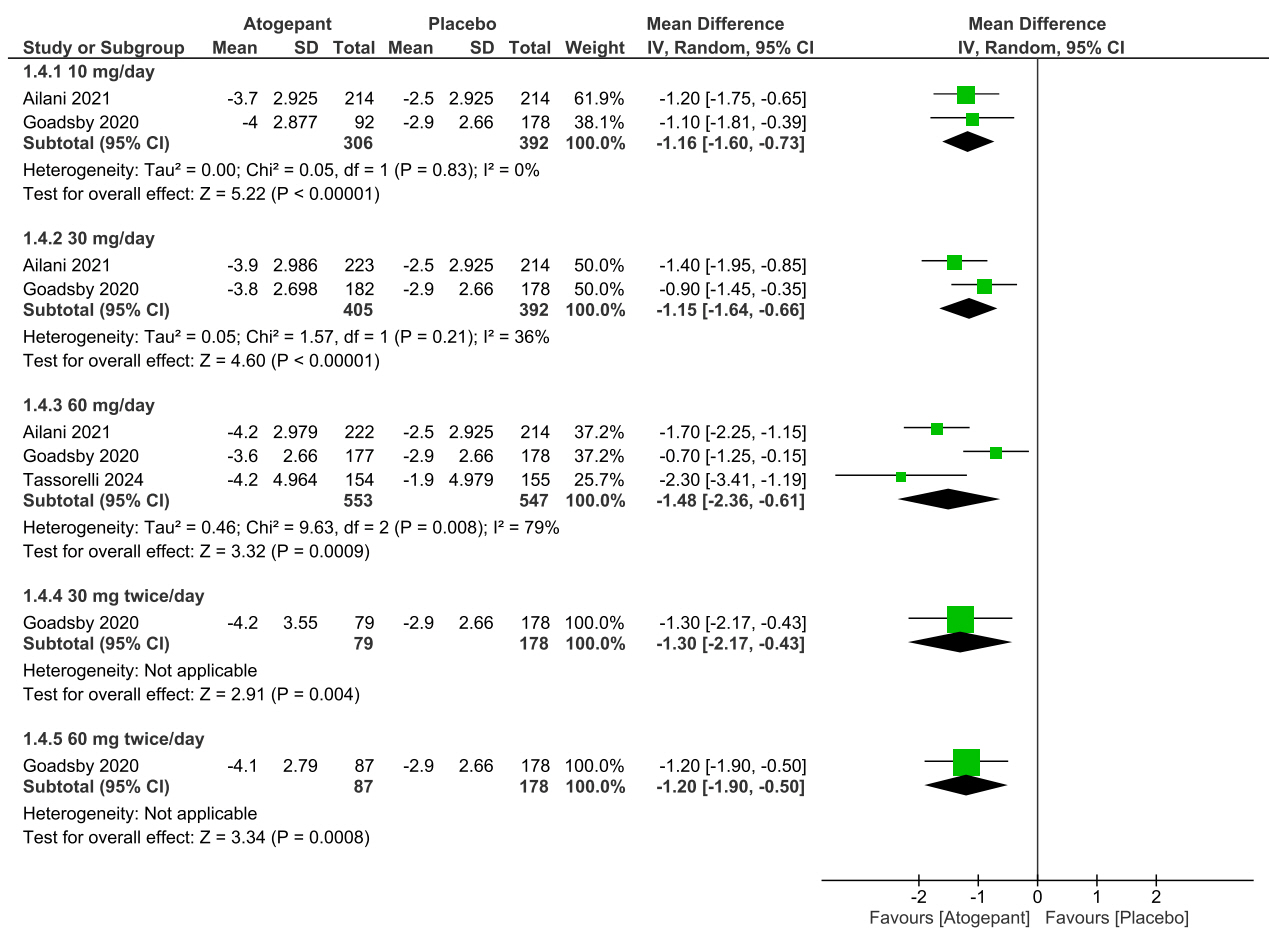
Forest plot of mean differences in monthly migraine days for various dosages of atogepant.
SD, standard deviation; IV, inverse variance; CI, confidence interval; df, degree or freedom.
• At 60 mg QD dosage: Three studies involving 553 patients in the atogepant group and 547 in the placebo group reported a significant reduction in migraine days for the 60 mg QD dosage (MD: –1.48 days, 95% CI: –2.36 to –0.61, p=0.0009), though with moderate heterogeneity (p=0.008, I2=79%) (Figure 2).
• At 30 mg BID and 60 mg BID dosages: In one study, atogepant significantly reduced monthly migraine days at both 30 mg BID (MD: –1.30 days, 95% CI: –2.17 to –0.43, p=0.004) and 60 mg BID (MD: –1.20 days, 95% CI: –1.90 to –0.50, p=0.005) (Figure 2).
• Combined doses vs. placebo: The pooled analysis of the four doses (10 mg QD, 30 mg QD, 60 mg QD, and 30 mg BID) across three studies (n=1,430 atogepant, n=547 placebo) demonstrated a significant reduction in monthly migraine days compared to placebo (MD: –1.40 days, 95% CI: –1.97 to –0.83, p<0.00001), with moderate heterogeneity (p=0.06, I2=64%) (Supplementary Figure 2, available online).
5. Analysis of ≥ 50% reduction in monthly migraine days
• At 10 mg QD and 30 mg QD dosages: Two studies13,15 showed that both 10 mg QD and 30 mg QD dosages resulted in a statistically significant number of patients achieving ≥50% reduction in monthly migraine days compared to placebo (RD: 0.23, 95% CI: 0.14–0.32, p<0.00001 for 10 mg; RD: 0.22, 95% CI: 0.05–0.38, p=0.01 for 30 mg). The number needed to treat (NNT) was approximately 5 for both doses (Figure 3).
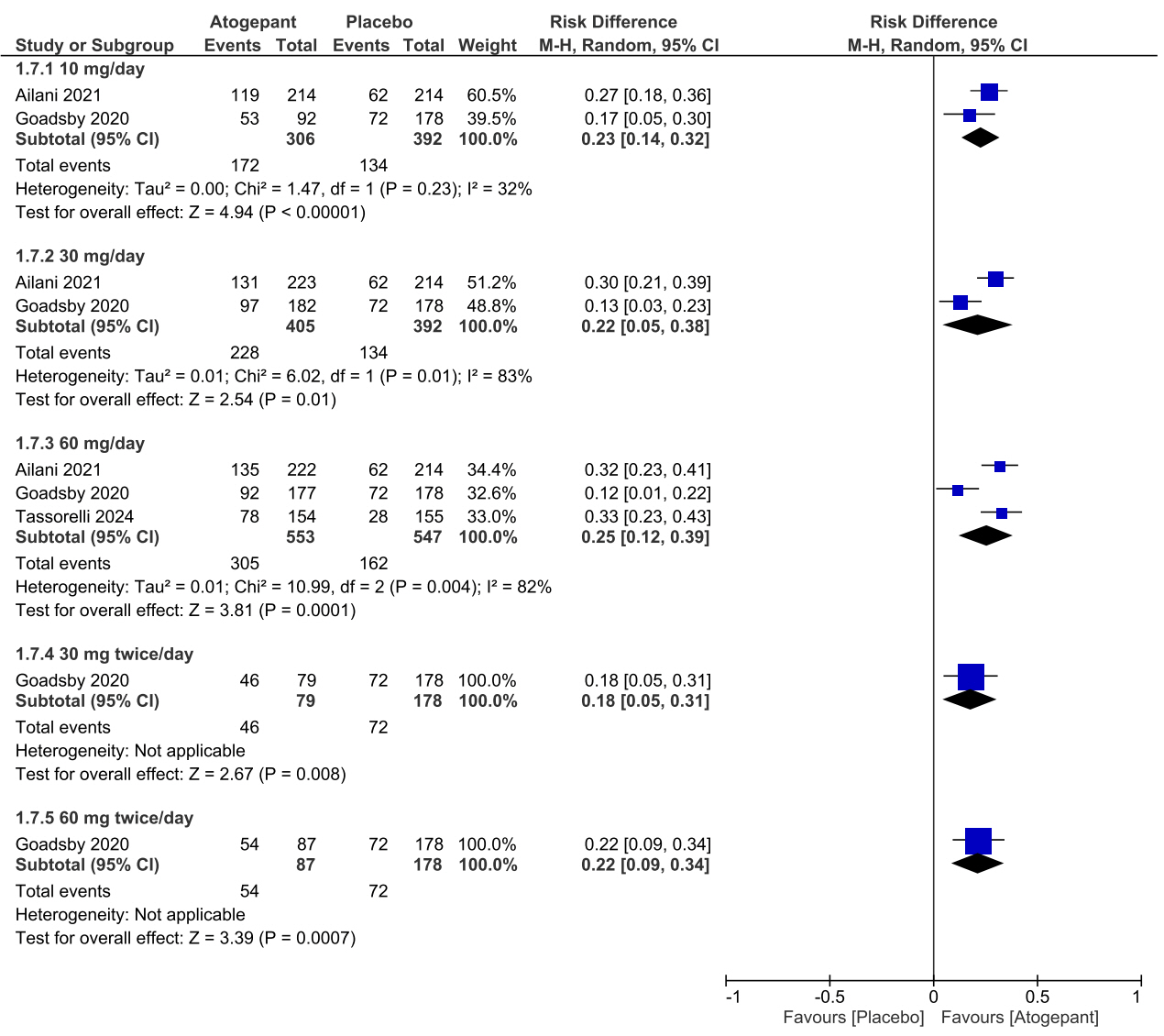
Forest plot of ≥50% reduction in monthly migraine days for various dosages of atogepant.
M-H, Mantel-Haenszel; CI, confidence interval; df, degree or freedom.
• At 60 mg QD dosage: In three studies, the 60 mg QD dose showed a significant effect (RD: 0.25, 95% CI: 0.12–0.39, p=0.0001), with an NNT of 4 (Figure 3).
• At 30 mg BID and 60 mg BID dosages: One study found that both 30 mg BID (RD: 0.18, 95% CI: 0.05–0.31, p=0.008) and 60 mg BID (RD: 0.22, 95% CI: 0.09–0.34, p=0.0007) significantly improved outcomes, with NNTs of 6 and 5, respectively (Figure 3).
• Combined doses vs. placebo: The pooled analysis revealed that atogepant across all doses significantly increased the proportion of patients achieving a ≥50% reduction in migraine days (RD: 0.26, 95% CI: 0.15–0.36, p<0.00001), with moderate heterogeneity (p=0.009, I2=79%) (Supplementary Figure 3, available online).
6. Mean difference in monthly headache days
• At 10 mg QD and 30 mg QD dosages: Atogepant at both 10 mg QD (MD: –1.40 days, 95% CI: –1.88 to –0.92, p<0.00001) and 30 mg QD (MD: –1.44 days, 95% CI: –1.90 to –0.98, p<0.00001) significantly reduced monthly headache days compared to placebo, with no significant heterogeneity (p>0.99, I2=0%) (Figure 4).
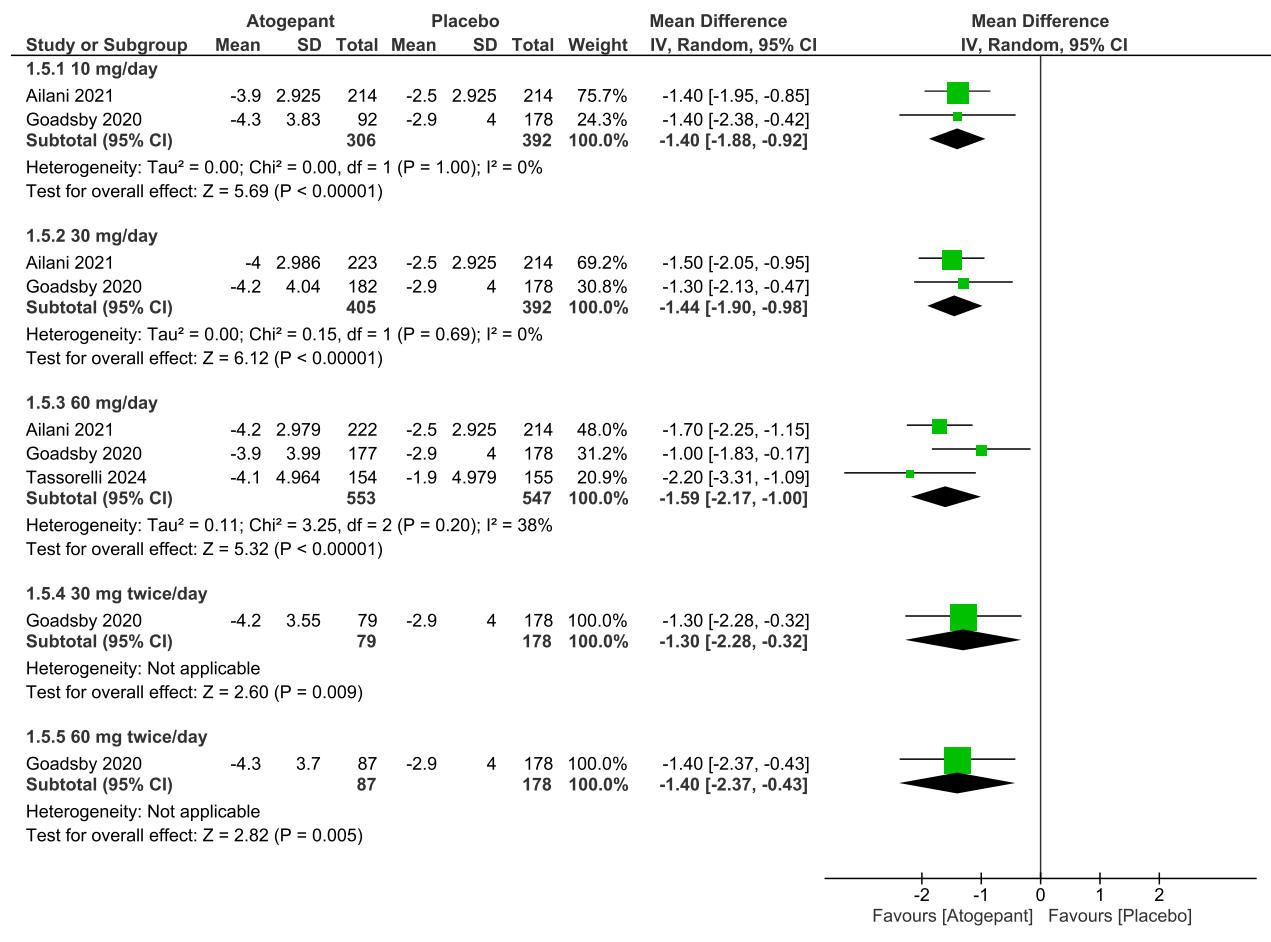
Forest plot of mean differences in monthly headache days for various dosages of atogepant.
SD, standard deviation; IV, inverse variance; CI, confidence interval; df, degree or freedom.
• At 60 mg QD dosage: The 60 mg QD dose also significantly reduced headache days (MD: –1.59 days, 95% CI: –2.17 to –1.00, p<0.00001), with low heterogeneity (p=0.20, I2=38%) (Figure 4).
• At 30 mg BID and 60 mg BID dosages: Atogepant significantly reduced headache days at 30 mg BID (MD: –1.30 days, 95% CI: –2.28 to –0.32, p=0.0009) and 60 mg BID (MD: –1.40 days, 95% CI: –2.37 to –0.43, p=0.0008) (Figure 4).
• Combined doses vs. placebo: The pooled analysis confirmed a significant reduction in monthly headache days for atogepant compared to placebo (MD: –1.52 days, 95% CI: –1.89 to –1.15, p<0.00001) with homogeneity of the data (p=0.35, I2=6%) (Supplementary Figure 2, available online).
7. Mean difference in monthly acute medication use days
Three studies3,15,23 reported data on acute medication use days.
• At 10 mg QD and 30 mg QD dosages: Both 10 mg QD (MD: –1.30 days, 95% CI: –1.74 to –0.86, p<0.00001) and 30 mg QD (MD: –1.40 days, 95% CI: –1.79 to –1.01, p<0.00001) significantly reduced acute medication use, with no heterogeneity (p>0.99, I2=0%) (Figure 5).
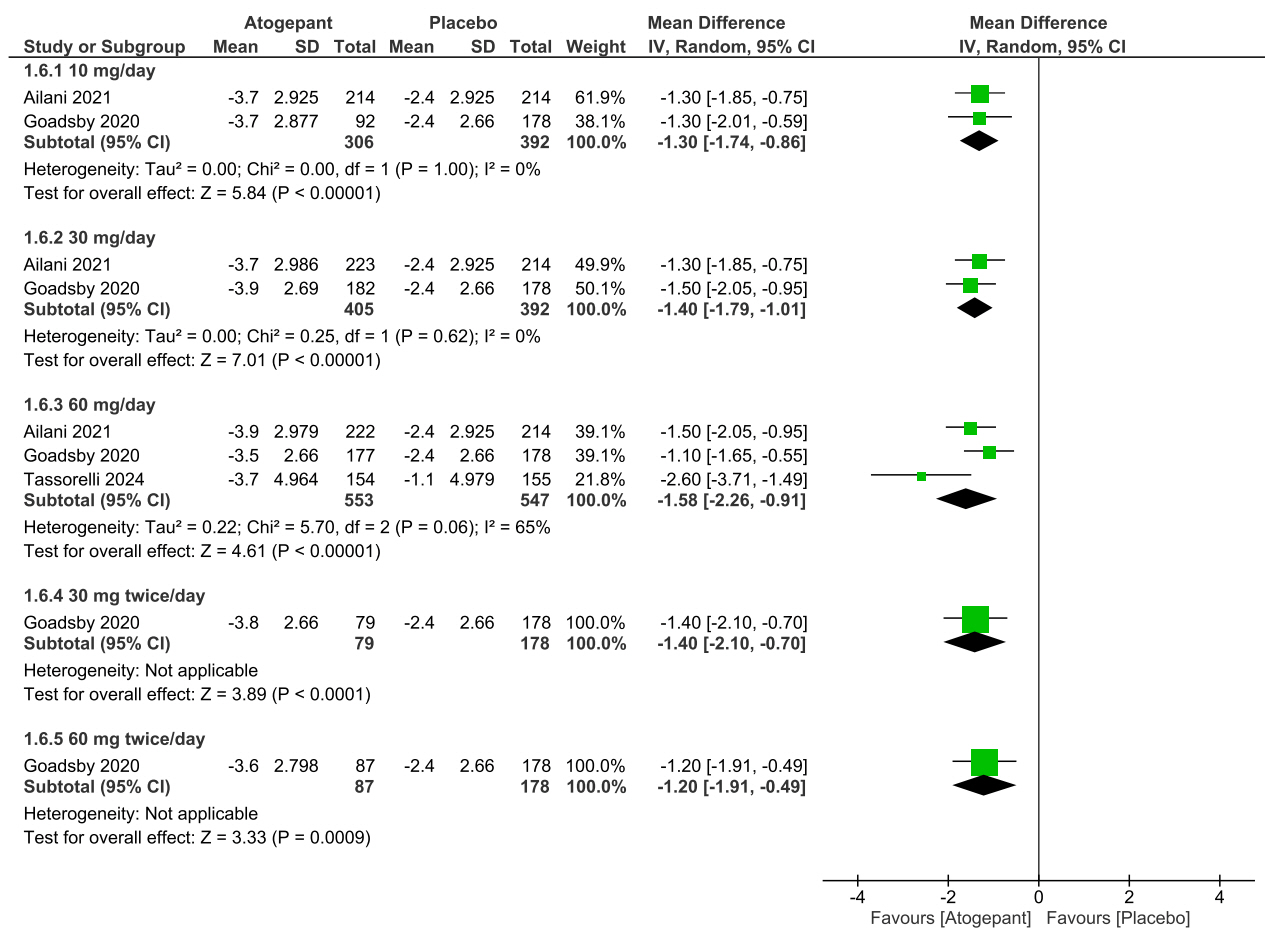
Forest plot of mean differences in monthly acute medication use days for various dosages of atogepant.
SD, standard deviation; IV, inverse variance; CI, confidence interval; df, degree or freedom.
• At 60 mg QD dosage: The 60 mg QD dose showed a significant reduction (MD: –1.58 days, 95% CI: –2.26 to –0.91, p<0.00001) with moderate heterogeneity (p=0.06, I2=65%) (Figure 5).
• At 30 mg BID and 60 mg BID dosages: Atogepant significantly reduced acute medication use days at 30 mg BID (MD: –1.40 days, 95% CI: –2.10 to –0.70, p<0.0001) and 60 mg BID (MD: –1.20 days, 95% CI: –1.91 to –0.49, p=0.0009) (Figure 5).
• Combined doses vs. placebo: The combined dose analysis confirmed significant reductions in medication use (MD: –1.54 days, 95% CI: –2.06 to –1.02, p<0.00001) with low heterogeneity (p=0.10, I2=57%) (Supplementary Figure 2, available online).
8. Dose comparison
Across the studies, there were no statistically significant differences in efficacy between the different dosages of atogepant (10 mg QD vs. 30 mg QD, 60 mg QD, 30 mg BID, or 60 mg BID) for the primary outcomes (p>0.05).
9. Adverse events
Atogepant was associated with a higher risk of treatment-related treatment-emergent AEs (TEAEs) (RR: 1.75, 95% CI: 1.36–2.25, p<0.0001), constipation (RR: 4.59, 95% CI: 2.29–9.22, p<0.0001), and nausea (RR: 2.16, 95% CI: 1.31–3.56, p=0.002) compared to placebo. No significant differences were found for other AEs, including SAEs and discontinuations (Figure 6).
10. Adverse events and dose dependence
Given that all dosages of atogepant demonstrated efficacy without significant differences between them, we performed a descriptive analysis focused solely on AEs reported at two or more dose levels. This approach aimed to evaluate potential dose-dependent trends in AEs, providing valuable insights for practitioners to better anticipate and manage AEs in their patients (Supplementary Table 1, available online).
• For ‘any TEAEs’, the incidence rates were similar across doses, with 56.7% at 10 mg QD, 57% at 30 mg QD, 54.45% at 60 mg QD, 60.4% at 30 mg BID, and 58.24% at 60 mg BID, showing no clear dose-dependent trend.
• ‘Treatment-related TEAEs’ revealed a potential increase specifically at 60 mg BID (26.4%), compared to lower doses (21.65% at 10 mg QD and 17.76%–20.9% for others), but no clear pattern in QD groups.
• For ‘SAEs’, incidence rates remained low, ranging from 0%–1.05%, with no apparent dose dependence.
• Similarly, for ‘TEAEs leading to treatment discontinuation’, rates varied (4.1% at 10 mg QD, 1.8% at 30 mg QD, and 2.325% at 60 mg QD) without a consistent trend.
• ‘Constipation’ demonstrated a dose-dependent increase in QD doses, rising from 6.05% at 10 mg QD to 7.15% at 60 mg QD, though BID doses showed a lower incidence (3.49%–4.4%).
• A strong dose-dependent relationship was observed for ‘nausea’, increasing consistently from 5.09% at 10 mg QD to 9.9% at 60 mg BID. Similarly, ‘fatigue’ exhibited a clear increase at higher doses, peaking at 9.9% with 60 mg BID, compared to lower QD doses (1.27%–3.35%).
• For ‘UTIs’, a peak incidence of 4.86% was reported at 30 mg QD, without a linear trend.
• ‘Nasopharyngitis’ showed a slight dose-dependent increase in QD doses, from 2.23% at 10 mg QD to 5.325% at 60 mg QD, while BID dosing rates remained inconsistent.
• ‘URTIs’ had variable rates (4.8%–6.97%) across doses with no clear trend.
• Other AEs, including ‘increased blood creatine kinase levels’ (6.97% at 30 mg BID), ‘sinusitis’ (1.8%–2.2%), ‘gastroenteritis’ (0.9%–2.2%), ‘influenza’ (1.4%–2.2%), and ‘sinus congestion’ (0.5%–1.7%), displayed minimal or inconsistent patterns across doses. Notably, ‘anxiety’ showed a slight increase at 60 mg QD (2.2%) compared to lower doses (0.4%–0.9%), while ‘somnolence’ (1.7%–3.2%) and ‘increased alanine aminotransferase levels’ exhibited no clear dose relationship.
In conclusion, dose-dependent trends were most apparent for ‘nausea’, ‘fatigue’, and ‘constipation’, particularly in higher doses, while other AEs showed inconsistent or minimal trends across the dose groups. The incidence for these three AEs is visualized in Figure 7.
DISCUSSION
Our meta-analysis demonstrated that atogepant at doses of 10 mg QD, 30 mg QD, 60 mg QD, 30 mg BID, and 60 mg BID significantly reduced the number of monthly migraine days and monthly headache days compared to placebo, highlighting its efficacy in migraine prevention. The dose-dependent reduction in monthly migraine days, ranging from –1.16 to –1.48 days, is consistent with other CGRP receptor antagonists, further validating atogepant as an effective preventive treatment for EM.
Moreover, atogepant was effective in reducing acute medication use days, particularly at higher doses, which is clinically relevant in reducing the reliance on rescue medications during migraine attacks. A ≥50% reduction in monthly migraine days was also observed in a significant proportion of patients, with RD ranging from 0.18 to 0.25 depending on the dosage, underscoring the dose-response relationship and therapeutic potential of atogepant.
The efficacy of atogepant across different dosages provides flexibility in tailoring treatment based on individual patient needs. The choice of dosage (10 mg, 30 mg, or 60 mg QD) should be guided by factors such as the frequency of migraine attacks, the presence of comorbidities, and the potential for drug interactions.25 Dose reduction or contraindication of atogepant should be considered in specific conditions, such as concurrent use of strong CYP3A4 inhibitors (e.g., ketoconazole) and in patients with severe renal or hepatic impairment.26 Higher doses, while more effective, may also be associated with a greater incidence of AEs, which must be weighed against the benefits for individual patients.
The safety profile of atogepant is an essential consideration in its clinical use. Our analysis revealed that treatment with atogepant was associated with a higher incidence of treatment-related TEAEs, constipation, and nausea, compared to placebo. However, no significant differences were observed for other AEs, including UTIs, URTIs, fatigue, nasopharyngitis, increased blood creatinine levels, or deaths. Additionally, there was no significant difference in treatment discontinuations due to AEs or in the incidence of SAEs between atogepant and placebo. These findings suggest that atogepant is generally well tolerated but warrants monitoring for gastrointestinal side effects, particularly in patients with pre-existing gastrointestinal conditions.
The introduction of atogepant into clinical practice could substantially improve patients’ quality of life by reducing the frequency of migraine attacks, decreasing medication use, and lowering healthcare costs associated with migraine care. This is particularly significant given the high disability burden of EM and the economic impact of migraine on healthcare systems.27-30
A unique aspect of our work is the comparative analysis of different atogepant doses to evaluate if there were statistically significant differences in efficacy across dosages. Importantly, when comparing doses directly, no statistically significant differences were observed in efficacy for any primary outcome. This finding suggests that the therapeutic effect of atogepant does not substantially increase with higher doses, indicating a potential plateau in dose-response. Clinically, this supports the use of lower doses, such as 10 mg QD or 30 mg QD, to achieve similar benefits while potentially minimizing the risk of AEs associated with higher dosages. By adhering strictly to Cochrane guidelines, we provided a methodologically sound and clinically relevant assessment of atogepant’s efficacy and safety profile in EM prevention.
Schwedt et al.31 reported that approximately 62% of participants receiving galcanezumab and 61% of participants receiving rimegepant achieved a ≥50% reduction in monthly migraine days, with no statistically significant difference between the two treatments.
In our meta-analysis, atogepant demonstrated a similar response, with 23% to 25% of participants achieving a ≥50% reduction in monthly migraine days depending on the dose, corresponding to an NNT of 4 to 5 across doses. These differences may reflect variations in trial populations, baseline characteristics, and endpoints assessed. The trial by Schwedt et al.31 primarily compared galcanezumab and rimegepant in a single trial setting, whereas our meta-analysis pooled multiple trials of atogepant, focusing on dose-response relationships and placebo-controlled outcomes.
Furthermore, Schwedt et al.31 highlighted that monoclonal antibodies, such as galcanezumab, often require subcutaneous administration, which can pose adherence challenges despite their efficacy. In contrast, atogepant’s oral formulation aligns with patient preferences for non-invasive options, offering a convenient alternative without compromising efficacy.
Rimegepant, another CGRP receptor antagonist with a half-life similar to atogepant (approximately 11 hours), supports a QOD dosing schedule as an alternative to daily dosing.32,33 This option could benefit patients with concerns about daily medication, offering greater flexibility without sacrificing clinical efficacy.
In conclusion, while direct comparisons between atogepant and CGRP monoclonal antibodies like galcanezumab are limited by differences in study design and methodology, our findings support atogepant as a flexible and effective preventive option for EM, particularly for patients seeking oral treatment alternatives. Further head-to-head studies would be valuable to establish the comparative effectiveness of these therapies.
1. Implications for clinical practice
Atogepant offers versatile dosing options that can be tailored to individual patient needs and treatment goals. In our analysis, we conducted a head-to-head comparison of different atogepant doses (10 mg QD, 30 mg QD, 60 mg QD, 30 mg BID, and 60 mg BID) across all efficacy outcomes, including reductions in monthly migraine days, headache days, and acute medication use. The results showed no statistically significant differences in efficacy between the various doses. This finding indicates that clinicians can prioritize dose selection based on individual patient preferences, tolerability, and clinical circumstances rather than relying on higher doses to achieve greater efficacy.
The 60 mg QD dose is the most effective option for patients requiring robust reductions in monthly migraine and headache days. It also minimizes acute medication use, making it particularly suitable for individuals with frequent migraines seeking potent preventive effects.
For patients prioritizing a balance between efficacy and tolerability or those sensitive to side effects, the 30 mg QD dose serves as an excellent starting point. This dosage effectively reduces migraine days and acute medication use while maintaining a favorable safety profile, making it an ideal choice for achieving preventive benefits with fewer AEs. The 10 mg QD dose provides an alternative for patients with milder symptoms or those initiating preventive therapy. While its impact on migraine and headache days is slightly lower than higher doses, it still offers meaningful reductions in attack frequency with minimal side effects. For patients requiring a more intensive approach, the BID dosing options—30 mg or 60 mg BID—offer additional flexibility. These regimens may benefit patients who do not achieve adequate relief with QD dosing or prefer split dosing throughout the day.
In summary, the 60 mg QD dose is optimal for maximum efficacy, while the 30 mg QD dose balances effectiveness and tolerability. Lower doses and BID regimens provide personalized options, making atogepant a flexible and patient-centered choice for migraine prevention.
2. Limitations and recommendations
This meta-analysis has several limitations that may affect the generalizability and reliability of the findings. First, the inclusion of only four RCTs represents a relatively limited evidence base, reducing the statistical power and precision of the estimates. Despite subgroup analyses to address this limitation, significant heterogeneity among studies, particularly regarding dosing regimens, raises concerns about the consistency of results. Variability in study designs, patient populations, and outcome measures further complicates direct comparisons.
Additionally, three of the four RCTs were conducted in the United States, limiting the findings’ external validity to non-United States populations. The under-representation of male and non-White patients, as noted by Tassorelli et al.,23 restricts the applicability of the results to more diverse demographics. Expanding future research to include broader patient populations is essential to enhance generalizability, especially in regions with differing healthcare systems and patient characteristics.
Another notable limitation is the lack of long-term safety and efficacy data, particularly for patients resistant to multiple migraine therapies. The current evidence focuses on short-term outcomes, leaving uncertainty about atogepant’s long-term impact, especially in patients with complex clinical profiles or comorbidities, which may influence its pharmacokinetics and safety.
Furthermore, questions remain about the relative advantages of oral CGRP antagonists like atogepant compared to injectable monoclonal antibodies targeting the CGRP receptor. While oral formulations offer convenience, issues of adherence, patient acceptance, and long-term preference require further investigation. Patient-centered research is crucial to better understand these factors and their influence on clinical outcomes.
Safety concerns, particularly gastrointestinal side effects such as reduced motility and constipation,15 also warrant attention. These AEs may affect patient quality of life and should be carefully monitored, especially in individuals with pre-existing gastrointestinal conditions.
Future research should prioritize long-term safety and efficacy studies, particularly in EM patients and those with complex treatment histories. Additionally, trials should include more diverse patient populations in terms of gender and ethnicity to improve generalizability. Investigating optimal dosing strategies and comparing atogepant to other CGRP-targeted therapies will also be essential to better define its role in the prevention and management of EM.
CONCLUSION
Atogepant demonstrates significant efficacy in reducing monthly migraine and headache days and decreasing acute medication use, making it a valuable option for EM prevention. Its selective CGRP receptor antagonism underpins its clinical benefits, though side effects such as constipation and nausea require careful monitoring. While evidence supports its short-term safety and efficacy, gaps remain regarding long-term use in EM and in diverse demographic groups. The under-representation of male and non-White patients highlights the need for broader, more inclusive research. Additionally, further studies should investigate optimal dosing strategies to refine atogepant’s clinical role and enhance its utility in the prevention of EM. Addressing these gaps will improve outcomes for diverse EM patient populations.
Notes
AVAILABILITY OF DATA AND MATERIAL
This systematic review and meta-analysis relied on publicly available data from previously published studies. The original research contributions utilized in this study can be accessed within the main article and supplementary materials.
AUTHOR CONTRIBUTIONS
Conceptualization: AMA, AA; Methodology: AMA, AA; Investigation: AMA, AM, MEM, HS; Data Curation: AMA, AM, MEM, HS; Formal Analysis: AMA, AA; Validation: AMA, AA, HS, AO; Writing–original draft: AA, SAA, HA, MTH, AO; Writing–review & editing: AMA, AA, HS, AO.
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING STATEMENT
Not applicable.
ACKNOWLEDGMENTS
We extend our gratitude to Meta-Analysis CRO for their invaluable contributions to this study, particularly in refining the analytical approach.
Supplementary Material
Supplementary materials are available from https://doi.org/10.62087/hpr.2024.0030.
Supplementary Table 1.
Summary of adverse events in atogepant and placebo groups
Supplementary Figure 1.
Risk of bias assessment using ROB-2 tool.
Supplementary Figure 2.
Analysis of mean monthly migraine days, headache days, and acute medication use days for combined atogepant dosages vs. placebo.
Supplementary Figure 3.
Analysis of ≥50% reduction in monthly migraine days for combined atogepant dosages vs. placebo.




