Alcohol-Induced Headache: A Narrative Review Based on Migraine Pathophysiology
Article information
Abstract
Alcohol-induced headache (AIH) is one of the most common headache experiences and is associated with a substantial socioeconomic burden; however, its pathophysiological mechanisms and clinical classification remain inadequately defined. Accumulating evidence indicates that AIH shares key biological pathways with migraine, particularly involving activation of the trigeminovascular system and calcitonin gene-related peptide (CGRP) signaling. Although currently available hangover remedies are supported by limited high-quality evidence, anti-CGRP treatments have emerged as a biologically plausible option for the situational prevention and acute treatment of AIH. A phenotype-based approach is therefore essential for achieving accurate diagnosis and effective management of alcohol-related headaches. Future well-designed clinical trials focusing on CGRP antagonists are warranted to address this common yet neglected disorder.
INTRODUCTION
Alcohol-induced headache (AIH) is one of the most common headache experiences in the general population, with a lifetime prevalence of approximately 72%.1 In a recent cross-sectional study of 347 Dutch adults, 8.1% of workers reported at least one episode of absenteeism per year due to alcohol hangover, and the estimated national annual cost exceeded €2.6 billion.2 Despite this substantial individual and societal burden, scientific interest in alcohol hangover and AIH has remained surprisingly limited.3 This neglect likely stems from a blend of cultural attitudes about drinking for leisure and the seemingly straightforward assumption that hangovers can be prevented by simply reducing alcohol consumption.
AIH has accompanied human history for as long as alcohol itself. Archaeological evidence from Israel has identified beer residues dating back approximately 13,000 years.4 In ancient Persian medical texts describing headache disorders over 9,000 years ago, AIH was reported as one of the most frequently mentioned headache phenotypes, ranking third overall.5 Yet, despite this long history, systematic attempts to classify AIH are relatively recent. The International Classification of Headache Disorders (ICHD) introduced the distinction between immediate and delayed AIH in ICHD-2, a framework that has been retained in ICHD-3.6,7 However, this classification has been occasionally questioned.8,9 As acknowledged within the ICHD itself, alcohol is one of the most common triggers of migraine.10,11 This creates an inherent diagnostic ambiguity. For example, if a patient with episodic migraine develops a bilateral pulsatile headache the day after drinking red wine, should this be classified as alcohol-triggered migraine or as delayed AIH? Furthermore, considering that alcohol consumption increases the production of nitric oxide (NO),12 it may also be classified as “Delayed NO donor-induced headache”. Although one of the core ICHD principles is that a diagnosis should be “not better accounted for by another disorder,” this criterion becomes difficult to apply when headache phenotypes overlap substantially.
In clinical practice, such headaches are often managed as an extension of migraine, for example with triptan treatment. This pragmatic approach is not without rationale. Accumulating evidence suggests that many forms of AIH share key pathophysiological mechanisms with migraine, including trigeminovascular activation, calcitonin gene-related peptide (CGRP) signaling, and neuroinflammatory processes.13 It should be emphasized, however, that alcohol avoidance or reduction remains the primary clinical recommendation for alcohol-related headache, and that pharmacological approaches discussed in this review are secondary and exploratory in nature.
In this narrative review we aim to interpret AIH through the framework of migraine pathophysiology. Due to the narrative nature of the review, formal systematic review reporting systems (e.g., PRISMA) could not be applied. Relevant literature was identified through targeted database searches and manual reference screening, based on the expertise of the authors. We discuss the mechanistic overlap between AIH and migraine, propose a phenotype-based approach to classification, and review current evidence on treatment and prevention, highlighting critical gaps for future research.
CLINICAL PHENOTYPES AND CLASSIFICATIONS
1. Immediate alcohol-induced headache (International Classification of Headache Disorders, 3rd edition code 8.1.4.1)
Immediate AIH, also referred to as “cocktail headache,” is defined by the ICHD-3 diagnostic criteria shown in Table 1.7 This headache can occur rapidly, within 3 hours, even after small amounts of alcohol (e.g., one or two glasses of wine). The pain is typically bilateral and pulsating and is aggravated by physical activity. Affected individuals often prefer to lie down, a behavior that may reflect difficulty maintaining cerebral perfusion pressure secondary to alcohol-induced cerebral vasodilation.14
2. Delayed alcohol-induced headache (International Classification of Headache Disorders, 3rd edition code 8.1.4.2)
Delayed AIH, commonly referred to as “alcohol hangover headache,” develops 5–12 hours after heavy alcohol consumption and is defined by the ICHD-3 criteria summarized in Table 2.7 Duration and severity of headache may correlate with the amount of alcohol intake.15 Patients with delayed AIH also frequently report orthostatic headaches. In contrast to immediate AIH, delayed AIH occurs as part of a broader hangover syndrome and is accompanied by a cluster of autonomic, gastrointestinal, sleep-related, and cognitive symptoms.16 A characteristic feature of delayed AIH typically emerges when blood alcohol concentration (BAC) is declining or has returned to near zero (Figure 1).17 Experimental studies suggest that a peak BAC of at least 0.1% is generally required for a clinically relevant hangover to occur.18 According to Widmark’s equation,19 this corresponds approximately to the BAC reached 90 minutes after a 60-kg adult consumes one bottle of soju.
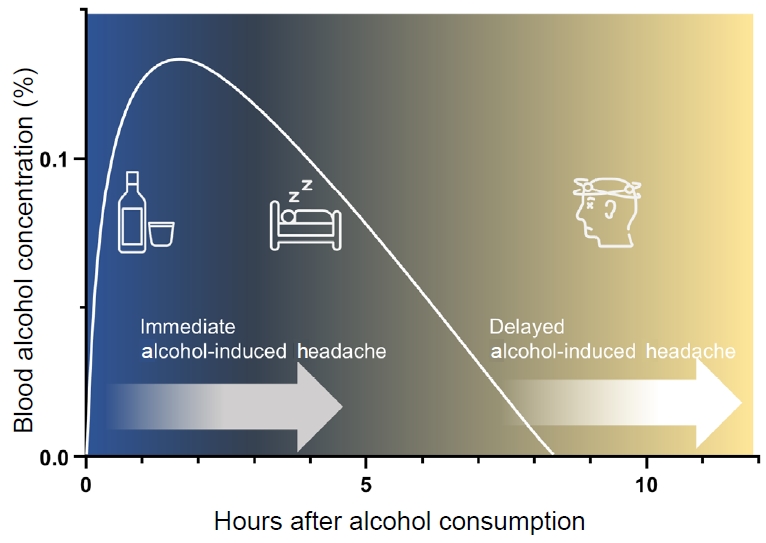
Schematic illustration of the temporal characteristics of alcohol-induced headache based on blood alcohol concentration.
The time-based distinction between immediate and delayed AIH in ICHD-3 is primarily operational and reflects consistent clinical observations rather than established mechanistic thresholds. To date, no definitive physiological evidence has validated a specific biological boundary underlying the 3-hour versus 5–12-hour cut-offs, and this classification should be interpreted as a pragmatic framework for clinical and research purposes.
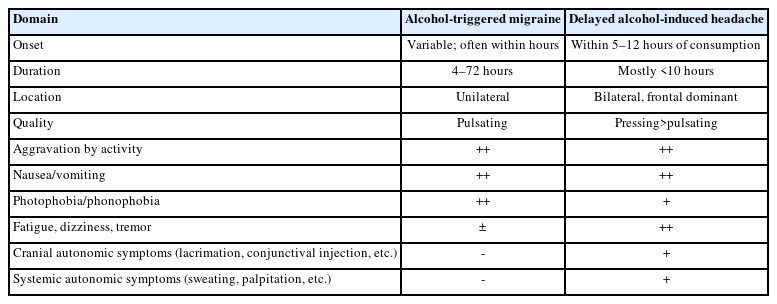
3. Distinguishing alcohol-triggered migraine from alcohol-induced headache
According to a cohort study in Korea, alcohol consumption was associated with an odds ratio (OR) of 2.5 for attack occurrence in episodic migraine.20 However, as discussed above, the current ICHD-3 framework does not clearly distinguish alcohol-triggered migraine from AIH in individuals with migraine. When patients present with recurrent headaches following alcohol consumption, we suggest that diagnosis should simultaneously consider temporal profile, headache phenotype, and individual susceptibility (Table 3). The most critical factor is whether the patient’s habitual migraine phenotype is faithfully reproduced after alcohol intake. This includes classical migraine features such as unilateral pain, nausea or vomiting, photophobia, and phonophobia. If these features are present, the headache should be classified as alcohol-triggered migraine. Conversely, when headache is accompanied by symptoms commonly associated with hangover, such as thirst, impaired concentration, apathy, diarrhea, anorexia, and autonomic symptoms, it should be classified as delayed AIH. However, despite this approach, many patients may exhibit overlapping or inconsistent clinical phenotypes. In such cases, we recommend assigning alcohol-triggered migraine as the primary diagnosis for clinical practicality, while interpreting non-headache symptoms and residual complaints as part of the hangover spectrum.
EPIDEMIOLOGICAL ASSOCIATIONS WITH PRIMARY HEADACHES AND SUSCEPTIBILITY
Evidence that alcohol precipitates headache can be found in numerous epidemiological studies. Paradoxically, many population-based studies consistently show that individuals with primary headache disorders consume less alcohol than those without headache. In a Norwegian study including 51,000 participants, headache prevalence increased 10%–20% in non-drinkers compared to moderate drinkers, across all age groups.21 Similarly, a Danish cross-sectional study of 46,000 individuals reported that weekly alcohol consumption was associated with a lower odds of migraine compared with abstinence (OR=0.55).22 This inverse association was replicated in analyses of the U.S. National Health and Nutrition Examination Survey (OR=0.54).23 These findings do not suggest that alcohol protects against headache. Rather, they indicate that headache sufferers are more likely to avoid alcohol.
When stratified by headache subtype, the association between migraine and alcohol becomes more pronounced. A recent meta-analysis demonstrated that alcohol consumption was not significantly associated with the risk of tension-type headache, whereas drinkers had a 1.5-fold lower likelihood of migraine compared with nondrinkers.24 Among patients with migraine, approximately 34%–35% report alcohol as a triggering factor.25,26 Interestingly, however, only a minority of migraine patients who abstain from or avoid alcohol, ranging from 3% to 25%, explicitly report headache as the primary reason for avoidance.26,27 This observation suggests that alcohol avoidance in migraineurs may occur both consciously and unconsciously, possibly reflecting learned or conditioned behavioral adaptation. Although alcohol avoidance can also be influenced by external and social factors such as religion, gender, and social norms, the consistent inverse association observed across diverse populations supports a prominent role of behavioral adaptation related to headache susceptibility.
Genetic susceptibility seems to play a critical role in AIH and hangover. A study analyzing 2,248 participants of twins found that over 40% of alcohol-related hangovers are determined by genetic factors.28 Inactive variants of aldehyde dehydrogenase 2 (ALDH2) are strongly associated with alcohol flushing, severe hangover symptoms, and headache.29,30 In a large Japanese cross-sectional study, ALDH2 deficiency demonstrated high sensitivity and specificity for alcohol flushing and hangover. Women and individuals exhibiting flushing reactions were significantly more likely to experience hangover headache. Notably, among individuals with migraine, those carrying inactive ALDH2 variants reported lower alcohol consumption and more pronounced adverse effects after drinking, supporting the presence of a gene–environment interaction in alcohol-related headache susceptibility.
Evidence regarding the role of specific alcoholic beverage types in triggering AIH remains limited. Fermented beverages generally contain higher levels of congeners produced during the fermentation process, which contribute to flavor and aroma but have also been implicated as potential contributors to hangover severity.9 Accordingly, some studies have suggested that red wine may provoke headache more frequently than white wine,31 whereas others have reported opposite or inconsistent findings.25
Importantly, no studies have systematically compared the severity of AIH across different beverage types under standardized BAC conditions. Moreover, most animal models of AIH rely on the administration of pure ethanol rather than alcohol beverages.14,32,33 Given the substantial heterogeneity in beverage preferences, drinking patterns and cultural contexts across populations and nations, the present review focuses primarily on the effects of alcohol itself rather than beverage-specific factors.
PATHOPHYSIOLOGICAL MECHANISMS LINKING ALCOHOL AND MIGRAINE
1. Central mechanisms: trigeminovascular activation and calcitonin gene-related peptide signaling
AIH was historically attributed primarily to peripheral vasodilation.34 However, accumulating evidence now indicates that activation of the trigeminovascular system and enhanced CGRP signaling represent central mechanisms directly linked to migraine pathophysiology.13,35,36 Migraine pain originates from activation of trigeminal afferents innervating the meninges and perivascular structures.37,38 Following alcohol consumption, ethanol is metabolized to acetaldehyde, which directly stimulates this system and activates trigeminal afferents.
Experimental animal models have demonstrated that gastrointestinal administration of alcohol activates members of the transient receptor potential (TRP) ion channel family, including transient receptor potential cation channel subfamily V member 1 (TRPV1) and Transient receptor potential cation channel, subfamily A, member 1 (TRPA1), leading to increased CGRP release.14,32 CGRP released from trigeminal nerve terminals acts on neurons and satellite glial cells within the trigeminal ganglion, promoting peripheral sensitization, while simultaneously activating second-order nociceptive neurons in the brainstem to induce central sensitization.37 These mechanisms provide a biological explanation for why AIH frequently manifests not only as pain but also with migraine-like extracranial features such as cutaneous allodynia and photophobia.32,39 Notably, these alcohol-induced effects are attenuated by CGRP antagonists, such as olcegepant, or by silencing receptor activity–modifying protein 1, a key component of the CGRP receptor complex, in animal model.32 Together, these findings suggest that anti-CGRP therapies may be effective in suppressing the clinical expression of AIH.
2. Peripheral mechanisms: nitric oxide signaling, vascular reactivity, and inflammation
Both animal and human studies consistently demonstrate that alcohol induces complex and bidirectional changes in NO signaling.12,39,40 Acute or low-dose alcohol exposure activates endothelial NO synthase, resulting in increased NO production and vasodilation. NO promotes cerebral blood flow while activating cyclic guanosine monophosphate signaling pathways that facilitate CGRP release from trigeminal nerve terminals.37 In contrast, chronic exposure to high-dose alcohol leads to depletion of NO bioavailability and antioxidant defenses through oxidative stress.12,33 Although this mechanism contributes to hypertension in chronic alcoholism, it may also offer insight into the paradoxically lower headache frequency reported in long-term heavy drinkers. Importantly, the delayed nature of NO-induced headache provides a plausible explanation for why delayed AIH is not strictly dose dependent and why headache frequently emerges during the declining phase of BAC.41
In parallel, alcohol consumption elicits systemic and central inflammatory responses. Elevated levels of cytokines, including interleukin-10, interleukin-12, and interferon-γ, as well as increased prostaglandin production, have been consistently observed during alcohol hangover.16,42 These inflammatory mediators are related to headache, nausea, and malaise and overlap substantially with pathways implicated in migraine-related neuroinflammation.43
3. Metabolic and behavioral modifiers
The metabolic consequences of alcohol consumption are broad and help explain why certain individuals are particularly prone to migraine. Alcohol increases the production of reactive oxygen species and reactive nitrogen species, inducing oxidative stress within the trigeminovascular system.44,45 This mechanism is well recognized in alcohol-related cardiovascular disease and may also be relevant to migraine biology. In migraine, thiobarbituric acid–reactive substances have been proposed as potential biomarkers, and preliminary studies suggesting therapeutic effects of antioxidants support a shared oxidative stress pathway linking AIH and migraine.44,46 Alcohol also inhibits diamine oxidase, the primary enzyme responsible for histamine degradation, thereby increasing endogenous histamine levels.47 Elevated histamine exerts vasodilatory effects and represents another potential mechanism contributing to migraine susceptibility following alcohol intake.43
Furthermore, alcohol disrupts sleep architecture and increases sleep fragmentation, while also elevating next-day anxiety levels.48,49 These behavioral and neuropsychological effects may lower the migraine threshold and place susceptible individuals in a state that favors headache generation.
CURRENT EVIDENCE AND THERAPEUTIC CONSIDERATIONS
1. Current evidence
To date, no clinical trial has specifically evaluated AIH as a primary outcome. Instead, most interventional studies have focused on the prevention or treatment of alcohol hangover as a broader syndrome.50 Based on randomized controlled trials, the following agents have demonstrated some efficacy in reducing hangover symptoms: clovinol,51 tolfenamic acid,52 pyritinol,53 Hovenia dulcis extract,54 L-cysteine combined with vitamins B and C,55 red ginseng,56 Korean pear juice,57 and HK-GCM-H01.58 Notably, four of these eight agents were evaluated in studies conducted in Korea. These compounds have been proposed to act through anti-inflammatory or prostaglandin-related pathways, or through antioxidant and metabolic mechanisms.50 In contrast, medications commonly considered anti-migraine agents, such as naproxen or propranolol,59,60 have not demonstrated benefit.
However, important limitations prevent us from extrapolating these findings directly to AIH. A 2022 systematic review of seven of the eight agents listed above found that all randomized trials were rated as having “very low quality” according to the GRADE (Grading of Recommendations Assessment, Development and Evaluation) methodology.50 This was largely due to small sample sizes and substantial selection bias.
A more fundamental limitation lies in the heterogeneity of hangover definitions and outcome measures. Most studies assessed hangover severity using composite scores derived from multiple symptoms, such as fatigue, thirst, sweating, and headache. These scales were often investigator-developed, insufficiently validated, and remain controversial in the literature.61 No single agent improved all symptom domains. Headache severity was usually evaluated using binary outcomes or simple numeric rating scales. These methodological issues substantially limit the applicability of existing hangover trials to AIH.
2. Interface with migraine treatment and the potential role of calcitonin gene-related peptide-pathway–based therapies
In clinical practice, when alcohol-triggered migraine is suspected, standard acute migraine treatments, such as triptans, are frequently used and appear effective. Most established acute migraine therapies can be considered in this setting, and there is currently no evidence suggesting clinically relevant drug–alcohol interactions for triptans.
In patients who experience migraine attacks with high probability following alcohol consumption, a strategy of mini-prophylaxis may be considered. For example, administration of a long-acting triptan, such as frovatriptan, at bedtime after alcohol intake may be conceptually analogous to mini-prophylaxis used for menstrual migraine.62 More recently, Lipton and colleagues63 introduced the concept of “situational prevention,” in which imegepant is used for prophylactic purpose during periods with increased risk of migraine attacks. AIH is a prototypical example of such a high-risk situation, in which pharmacological prevention may be considered in the setting of anticipated alcohol exposure, such as wine consumption during travel.
Although current evidence remains limited, CGRP-targeted therapies represent the most biologically plausible candidate for AIH prevention from a migraine-based perspective.13 Rimegepant, a CGRP receptor antagonist with both acute and preventive indications, is particularly attractive, as it may theoretically provide benefit in alcohol-triggered migraine as well as in immediate and delayed AIH.64 Nevertheless, important safety considerations remain. While imegepant has demonstrated minimal hepatotoxicity with short-term use,65 its safety profile when administered in the context of recent alcohol consumption, including repeated or situational use, remains insufficiently studied.
3. Key considerations for future clinical trials of calcitonin gene-related peptide antagonists in alcohol-induced headache
Several methodological issues must be addressed in future trials evaluating CGRP antagonists for the prevention or treatment of AIH. First, reproducibility of AIH must be ensured. AIH exhibits marked interindividual variability, and even within the same individual, identical alcohol exposure may or may not result in headache. A randomized, crossover trial design would therefore be optimal to control for this variability. Second, phenotypes of AIH should be clearly distinguished rather than pooled. For example, trials should separately evaluate migraineurs with reproducible alcohol-triggered migraine and non-migraineurs with reproducible delayed AIH. Third, alcohol exposure must be standardized. Target exposure should be defined by BAC rather than by beverage volume alone, for example aiming for a peak BAC of ≥0.1%. Verification can be achieved using breath analyzers or blood sampling. Fourth, confounding hangover-related factors, including sleep quality, mood, gastrointestinal symptoms, and hydration status, should also be measured and controlled. Finally, ethical considerations exist. Investigators must carefully assess whether study designs inadvertently encourage alcohol consumption and must adhere strictly to institutional review board standards regarding participant safety.
CONCLUSIONS
AIH is more than a simple consequence of alcohol consumption; it is a complex neurological phenomenon that closely intersects with migraine biology. By understanding AIH, clinicians can move beyond reactive hangover management toward a more proactive, mechanism-based therapeutic approach. The integration of situational prevention strategies and the potential application of CGRP antagonists offer promising avenues for patients suffering from recurrent AIH. Continued research with standardized methodologies will be crucial to bridge the gap between bench evidence and clinical practice.
Notes
AVAILABILITY OF DATA AND MATERIAL
Not applicable.
AUTHOR CONTRIBUTIONS
Conceptualization: WSH; Writing–original draft: WSH; Writing–review & editing: WSH.
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING STATEMENT
Not applicable.
ACKNOWLEDGMENTS
Not applicable.